They weren't actually that fast, that's the issue, people keep comparing it to vaccines from the 50's, the way it's down now vs then is quite different, plus e.g. J&J's version is based on their Ebola one where the method of delivery (in regards to aednovirus, disabled virus) was already in trials/tests since 2014, was approved in June 2020.
The only thing that really changed was method of delivery in regards to mRNA (so Pfizer and Moderna) which already had decades of research and has had human trials since 2011, and the biggest issue is that people don't understand that there are differing levels of complexity for virus' and that there was already a vaccine for SARS-CoV-1 that could be used as a base.
If scared, get the J&J if you can, most similar to what's been done before and method of delivery did full trials etc. for ~6 years already, but the Pfizer one again is not really that new, it's been tested for a long while and e.g. mRNA is there for ~2 weeks before being destroyed by the body, so you'd have seen side effects by now considering that it's been 7 months since the start of the mass vaccinations.
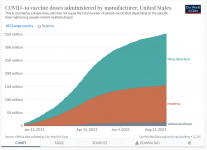
View attachment 27912
That's US data, you can pick whichever country you like:
https://ourworldindata.org/covid-vaccinations#which-vaccines-have-been-administered-in-each-country
Just ignore the Chinese data, so remove 1.8bn from total administered (~30%), as that's Sinovac.
Most of the delay for full approval always comes from the admin involved, as it's a specific handful of people that need to review tens of thousands of pages of data, that's the bit that was skipped (as in they had a larger team that reviewed all the prelim data etc. and as on-going), and that is usually 6 months to 1 year, Pfizer is set to get full approval some time next month.
J&J, Moderna, and Pfizer completed Phase 3 trials btw (so large clinical (human) trials), all of those were started and ended 2020's, so issues would have cropped up by now as vaccine issues 99% of the time are in the first 3 months.